Looking to expand your contract manufacturing activities in Latin America?
Fresenius Kabi Contract Manufacturing Services in Latin America
July 31, 2020Fresenius Kabi Contract Manufacturing core competencies allow our partners to have access to trained and professional people, state-of-the-art production facilities and capabilities in Latin America thus simplifying product´s path to market while meeting partner´s manufacturing needs.
Fresenius Kabi has provided contract manufacturing services for decades and is committed to further expanding these activities in Latin America. Spread across four production sites in Latin America, compliant with cGMP and local standards, our operations are equipped to become our partners´ manufacturing platforms. Fresenius Kabi constantly invests in its manufacturing sites to maintain them at the cutting edge of technology and staffed with well-educated and highly motivated people.
Fresenius Kabi Contract Manufacturing core competencies allow our partners to have access to trained and professional people, state-of-the-art production facilities and capabilities in Latin America thus simplifying product´s path to market while meeting partner´s manufacturing needs. Fresenius Kabi has an established quality, regulatory and supply-assurance track record, along with robust manufacturing expertise. Our portfolio offers products that are synonymous with quality, consistency and reliability.
The customized approach and flexibility allows the opportunity to offer tailored solutions for unique, specialized requirements. Once the development work is complete, we will formulate, fill/finish, label, and package your product to your specifications.
Fresenius Kabi Contract Manufacturing locations in LAM
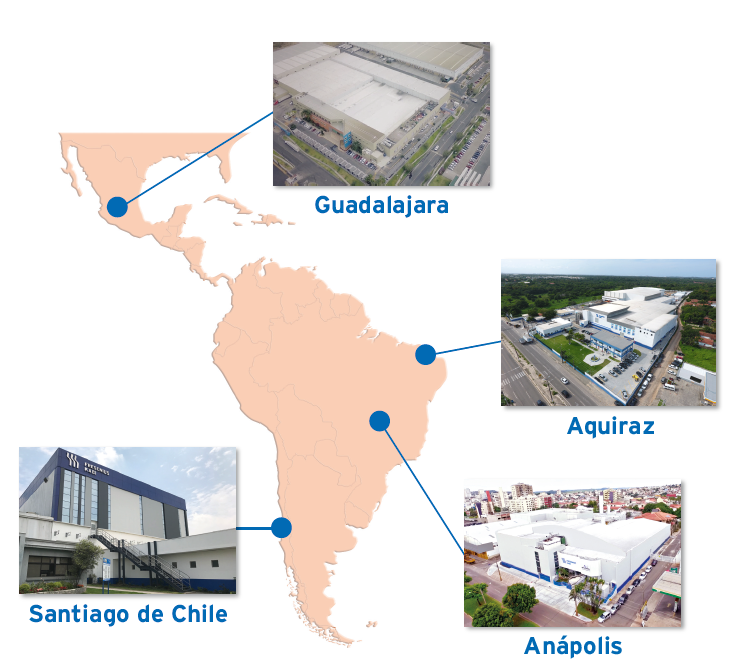
Anápolis (Brazil)
The production site in Anápolis (state of Goiás), located in the second biggest hub of pharmaceutical industry in Brazil, was founded in 1994 as a specialist for small volume parenterals and acquired by Fresenius Kabi in 2014. The facility has 6 dedicated production areas according to technologies used and therapeutical class of the final product. The plant produces a variety of small volume parenterals, i.e. liquids (glass ampoules 1-10 ml) and powders (glass vials 0.1-4.5 g) through aseptic processing or final sterilization focusing on Antibiotics, Glucocorticoids, Analgesics and Catecholamine. The facility employs about 500 people and has its own quality, logistics and administration departments including an onsite warehouse. The site is GMP certified by Anvisa and obtained a GAFO (Grupo de Avaliação e Qualificação de Fornecedores/ Evaluation and Qualification Group for Suppliers) certification.
Aquiraz (Brazil)
One of our Brazilian production sites located in Aquiraz, in the state of Ceara, was founded in 1985 as a local company, and acquired by us in 1997. The plant produces Standard and IV Solutions in Plastic Bottles (50-1000 ml). The site has its own quality, maintenance, logistics, administration departments and warehouses for finished products and raw material storage. The facility has a highly qualified staff of about 600 employees and its products are produced to serve the Brazilian market. The site is GMP certified by Anvisa and ISO 9001 certified.
Santiago de Chile (Chile)
Operations in Santiago de Chile started in a small pilot plant making Standard Solutions in plastic bottles. Currently the plant produces a variety of products including generic IV Drugs in a large number of different containers such as freeflex® bags and plastic ampoules ranging in volume from 2 ml to 5000 ml. Furthermore, a high-tech integrated line for filling glass ampoules ranging in volume from 1 to 10 ml and glass vials ranging in volume from 1 to 20 ml within aseptic process or with final sterilization completes the capability of our Chilean site. Currently our “Laboratorio Sanderson” production site in Chile employs around 850 employees, and covers more than 32,000 square meters area. The production site in Santiago supports Fresenius Kabi´s business with a variety of containers and a wide range of products such as Standard Solutions, Anesthetics, Analgesics and Anticoagulants delivered to almost all Latin American countries and European non-profit organizations. The site is GMP certified by Anvisa and ISO 9001 certified. Additionally it holds GMP certifications of the local Chilean ministry of health as well as from Peru (DIGEMID) and Colombia (INVIMA).
Guadalajara (Mexico)
The production site in Guadalajara was founded as “Laboratorios Alpha” and acquired by us in 1996. In 2004 the site was completely renovated and since then the plant covers an area of 32,000 square meters (shared with our sister company Fresenius Medical Care). The plant produces Standard Solutions and generic IV Drugs in Blow-Fill-Seal Plastic Bottles (ranging in volume from 50 to 1000 ml) and in PVC bag containers (from 50 to 300 ml). All our IV products are terminally sterilized. The plant also has a line producing enteral nutrition products; i.e., Oral Powders in plastic containers and sachets, as well as Sip Feeds in aluminum cans. This production site is focused on providing products for the local Mexican market as well as for Peru, Argentina, Ecuador and further markets in the Central American region. Guadalajara is considered a commercial and financial center with excellent infrastructure and great connectivity to the rest of the country. The site is GMP certified by INVIMA (Colombia) and DIGEMID (Peru) and has an ISO 9001 certification.
